Tears of the rotator cuff are one of the most common reasons for shoulder pain and loss of function. Rotator cuff tears can result from an injury (Acute Tears of the Rotator Cuff: Why seek Help Immediately?). More often, the rotator cuff tendons wear over time (A Few Common Questions About Rotator Cuff Tears: How Do They Happen? Do They Heal? Do They Get Bigger? Why?). For reasons that are not clearly understood, some individuals with torn cuff tendons do not even know they have a tear! …, but many people with a torn rotator cuff experience shoulder pain, weakness, and other limitations.
When a chronic rotator cuff tear is the reason for pain and loss of function, a good program of physical therapy oftentimes will eliminate pain and improve function; occasionally, a local injection with corticosteroids (or an alternative anti-inflammatory) is needed as well. Unfortunately, some people do not get better with physical therapy, and at that point surgery is recommended.
Repair or fixation of the torn tendons to bone is the most common surgery recommended to individuals with a rotator cuff tear that either happened because of a substantial injury or has not responded to physical therapy (Arthroscopic Repair of the Rotator Cuff: How is it Done? What to Expect?). However, sometimes the tear is so bad that either it cannot be repaired surgically, or it has a very low chance of healing if repaired. Shoulder experts call these tears Functionally Irreparable Rotator Cuff Tears (FIRCT). These are factors that contribute to the irreparability of a rotator cuff tear:
- Very large tear size
- Majority of muscle replaced by fat
- Muscle atrophy
- Short tendon stump
- Stiff tissues

What are the options when a rotator cuff tear is irreparable?
Physical therapy can still work for certain patients with FIRCTs. However, some individuals with irreparable cuff tears do need surgery. Surgical procedures considered for irreparable cuff tears are listed below:
- Debridement (cleaning of the joint, smoothing of bone and ragged tendon edges)
- Partial repair (repair whatever portions of the tendon can be fixed)
- Tendon grafting or augmentation
- Reconstruction of the superior capsule of the shoulder
- Transfer of other healthy tendons to replace the torn rotator cuff (Transfer of the Lower Trapezius: An Option to Consider for Bad Rotator Cuff Tears that Cannot Be Fixed)
- Replacement of the shoulder with a reverse prosthesis (Reverse Shoulder Replacement: What You Need to Know)
- Implantation of an inflatable balloon
What is the subacromial balloon made of?
The only balloon approved in the United States for implantation in the shoulder is the InSpaceTM balloon, initially developed by OrthoSpace and currently owned and distributed by Stryker corporation. The InSpaceTM balloon is a co-polymer that combines the toughness of poly-L-lactide (70% of the composition) with the elasticity of e-caprolactone (30% of the composition). Some abbreviate poly-(L-lactide-co-e-caprolactone) as PLCL. This polymer is not toxic, does not elicit inflammation, and is biodegradable: the human body eliminates its material over time. PLCL resists cyclic pressure and retains its tensile strength even while being resorbed.

Once manufactured, the InSpaceTM balloon is folded into a cylinder and placed inside a protective cannula. At the time of surgery, the folded balloon is deployed in between the humeral head and acromion. Saline is then injected inside the balloon to inflate it before being sealed.
How does the subacromial balloon work?
In the presence of an irreparable rotator cuff tear, the ball of the shoulder (humeral head) migrates up and rubs against the acromion bone. In these circumstances, several factors may contribute to pain and loss of function: (1) the torn rotator cuff muscles cannot power the shoulder, (2) the mechanics of the joint are poor with such a high-riding humeral head, no longer aligned with the socket, (3) there is rubbing or friction between humeral head and acromion, and in some instances (4) the articular cartilage of the joint can experience progressive wear with arthritis.

Placement of an inflated balloon in the subacromial space is believed to improve pain and function two ways: (a) it does not allow the humeral head to rub with the acromion, and (b) the humeral head is returned to a more normal position across the glenoid socket, which improves the mechanics of the joint. However, it is not clear why individuals experience pain relief and improved function well after the balloon has already resorbed. The theory is that temporary physical centering of the humeral head across the socket and improvements in pain allow patients to strengthen the rest of the intact muscles round the shoulder so that they can permanently compensate for the torn rotator cuff.
The balloon can also be considered as an add-on for shoulders undergoing rotator cuff tear. There are two mechanisms that explain why adding a balloon can be beneficial when performing a cuff repair. First, by keeping the humeral head centered across the glenoid socket, the repaired cuff tendons are in a better mechanical position, they cannot rub against the acromion (which could lead to retear), and the rest of the shoulder structures are in a better mechanical position as well. Secondly, the pressure of the inflated balloon compresses the repaired tendons to bone further, potentially increasing healing.
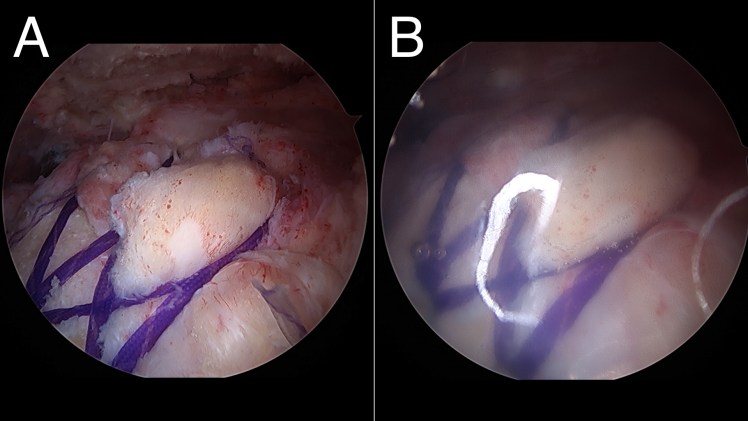
When is a balloon the best option for a FIRCT?
As mentioned above, there are several surgical procedures that may be considered for FIRCTs. The Food and Drug Administration has approved the InSpaceTM balloon for individuals that are (1) 65 or older with (2) an irreparable rotator cuff tear, (3) no or minimal arthritis, and (4) an intact subscapularis tendon. However, there are younger patients that may also be good candidates for a balloon. These are our current indications for implantation of a subacromial balloon:
- Combined with debridement, acromioplasty and/or biceps tenotomy or tenodesis
- Painful FIRCT
- No or minimal glenohumeral arthritis
- Relatively preserved active motion
- No major weakness in internal rotation or external rotation
- Intact or reparable subscapularis tendon
- Combined with rotator cuff repair
- Static proximal humeral head migration, or
- Risk factors for incomplete tendon healing
Patients with severe arthritis in the presence of a FIRCT are better candidates for a reverse shoulder arthroplasty (Reverse Shoulder Replacement: What You Need to Know). When the main limitation is severe loss of strength in external rotation, tendon transfers are probably best (Transfer of the Lower Trapezius: An Option to Consider for Bad Rotator Cuff Tears that Cannot Be Fixed).
How is the procedure performed?
Balloon implantation is performed under arthroscopic visualization. A small skin incision is used to introduce a camera in the shoulder region. A separate small skin incision is used not only to implant the balloon, but also to clean inflamed or degenerated tissue and performed other procedures if needed.

The distance between the top of the glenoid socket and the lateral humerus is measured to determine whether to open a small, medium, or large balloon. The balloon is then opened and inserted into the subacromial space protected by the outer cannula. Once the cannula is removed, saline is injected into the balloon to fully expand it, and a small amount of saline is then retrieved to leave the balloon at just the correct size. The balloon is then sealed and the inserter is removed.
The following table summarizes recommended inflation volumes for InSpace:
| Measured distance | Size/part number | Maximum inflation volume | Recommended final volume |
| < 40 mm | Small (0130) | 15 – 17 cc | 9 – 11 cc |
| 40 – 50 mm | Medium (0131) | 22 – 24 cc | 16 – 16 cc |
| > 50 mm | Large (0132) | 40 cc | 23 – 25 cc |
What about aftercare?
When other procedures (rotator cuff repair, biceps tenodesis) have been performed, the program of protection and therapy after surgery is determined by the needs of those procedures. When the balloon is inserted as a stand-alone procedure (or combined with debridement, acromioplasty and/or biceps tenotomy), a sling is recommended for two weeks, and therapy is started at that time.
Does it work, really?
When the balloon was developed in Israel and first used in Europe, many were skeptical about how effective a resorbable spacer device would be. Interestingly, several studies have reported good outcomes with balloon implantation. In fact, the study performed to request FDA clearance confirmed over 85% success in patients over 65 that met all other criteria for inclusion in the study. However, true high-quality long-term outcomes are still unknown.
Is it covered by Medicare?
Not at this point as an isolated procedure, but may be covered when associated with other procedures.
what other procedures might be used to get Medicare approval for the In Space procedure? This is a 73 year old woman with a shoulder rotary cuff problem.
The FDA gave approval for this procedure at this age..
The FDA has approved this procedure for patients 65-year-old and older. At Mayo Clinic we are about to start a study in which the balloon will be provided for free if the patient enrolls in the study. This study will probably kick off in November.
My name is Miguel Davis I am a 58 year old male. I was interested to see if I would be a candidate for the Subacromial balloon procedure and if so, would this be a viable means for me to move forward.
The below injury occurred while bench pressing…
The MRI which was obtained on 11/28/2022 and I have not had anything done yet on the injury.
ROTATOR CUFF:
. Supraspinatus tendon: Complete tear of the supraspinatus tendon with retraction to the level of the glenohumeral joint measuring approximately 4 cm.
. Infraspinatus tendon: Complete tear of the infraspinatus tendon with retraction to the level of the glenohumeral joint measuring approximately 4.2 cm.
. Subscapularis tendon: High-grade partial-thickness articular surface tear of the subscapularis tendon superior fibers. Background of severe tendinosis.
. Teres minor tendon: Intact.
LONG HEAD OF BICEPS: Complete tear of the long head biceps tendon above the groove. Residual portion of the long head biceps tendon is visualized within the tendon sheath.
The long head of the bicep injury occurred 5 or 6 years ago and I never had it repaired. I have been able to function fairly normally after this injury still able to work out curl substantial wieght etc…
My husband is going to have this done as he has re-torn his supraspinatus at the anchor site from his first surgery. I’ve been doing a lot of research and this article seems to be the most informative I have found. We are curious how it will help him long term. Hopefully he gets relief, as he has been in pain since his first tear in December 2019 and has already had 2 surgeries.